| Media Relations: | Investor Relations: |
|---|---|
| 908-989-0726 | 908-981-5560 |
| Email: [email protected] | E-mail: [email protected] |
Sanofi Delivers Q1 2015 Business EPS Growth of 2.6% at CER and 12.8% on a Reported Basis
Paris, April 30, 2015 / PRNewswire / — Sanofi (NYSE: SNY; EURONEXT: SAN)
| Q1 2015 | Change (reported) | Change (CER) |
|
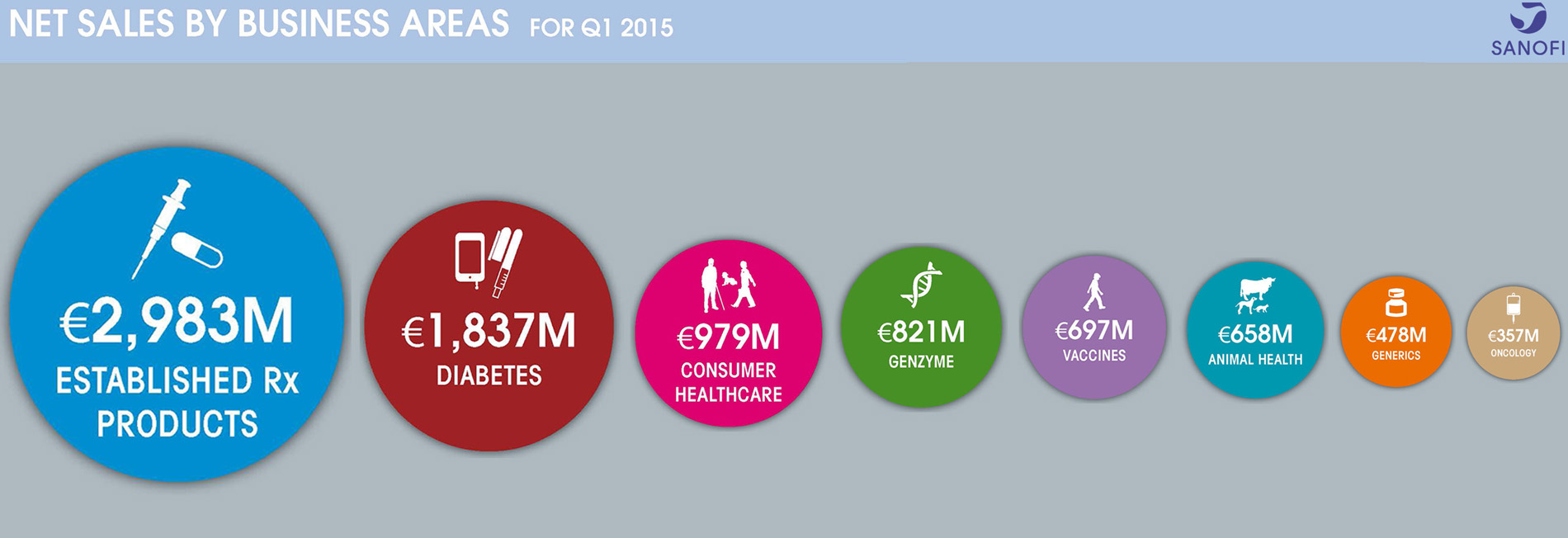
| Net sales | €8,810 m | +12.3% | +2.4% |
| Business net income(1) | €1,726 m | +11.6% | +1.6% |
| Business EPS(2) | €1.32 | +12.8% | +2.6% |
In order to facilitate an understanding of operational performance, Sanofi comments on the business net income statement. Business net income(1) is a non-GAAP financial measure. (2)(EPS) Earnings Per Share
Sanofi Chief Executive Officer, Olivier Brandicourt commented:
“Sanofi had a good start to 2015. Our businesses provide a solid foundation for our new-product cycle. Sanofi’s recent launches along with the ongoing regulatory reviews and planned submissions before year-end will drive future growth. At this important time for the company, my primary focus will be on maximizing the value of this innovative product portfolio and further establishing Sanofi as a leading biopharmaceutical company.”
Sanofi growth driven by Genzyme and Merial
- Group sales increased 2.4% (+12.3% on a reported basis) to €8,810 million
- Slightly lower Diabetes sales (-3.2%) reflects expected pricing impact on Lantus® in the U.S.
- Genzyme delivered 30.9% growth mainly driven by Aubagio®
- Animal Health recorded a strong quarter (+13.5%) driven by performance of NexGard®
- Vaccines (-4.6%) declined due to expected delay in Southern Hemisphere influenza campaign
- Emerging Markets sales up 7.3%
Solid financial performance
- Business net income(1) grew 1.6% at CER (up 11.6% on a reported basis) to €1,726 million despite launch investments and U.S. Lantus® pricing impact
- Business EPS(2) was up 2.6% at CER to €1.32 and increased 12.8% on a reported basis
Significant achievements with new product launches
- Toujeo® was launched in the U.S. and approved in EU
- Cerdelga® approved in Europe and in Japan
- Lemtrada® sales benefited from market introduction in the U.S.
- Dengue vaccine rolling submission initiated in 6 endemic countries in Asia and Latin America
- Results of ELIXA cardiovascular safety with lixisenatide support U.S. resubmission in Q3 2015
Further progress in R&D
- Phase III study of dupilumab in moderate-to-severe asthma initiated
- Phase IIb of the IL4/IL13 bi-specific mAb in idiopathic pulmonary fibrosis started
R&D Update
Regulatory updates since the publication of the 2014 results on February 5, 2015 include the following:
- In April, the FDA granted Fast Track designation to GZ/SAR402671, Genzyme’s oral substrate reduction therapy, for the treatment of Fabry disease.
- In March, Quadracel® (Diphtheria, tetanus, pertussis, polio vaccine) was approved in the U.S. in children 4-6 years of age.
- In March, Cerdelga® (eliglustat), an oral treatment for certain adults living with Gaucher disease type 1, was approved in Japan and in February in Australia.
- In February, the U.S. Food and Drug Administration (FDA) approved Toujeo®, a next generation basal insulin used to treat adults with type 1 and type 2 diabetes. In April, Toujeo® was also approved in EU.
At the end of April 2015, the R&D pipeline contained 37 projects (excluding Life Cycle Management) and vaccine candidates in clinical development of which 12 are in Phase III or have been submitted to the regulatory authorities for approval.
2015 Guidance
- Sanofi continues to expect 2015 Business EPS(1) to be stable to slightly growing versus 2014 at constant average exchange rates, barring major unforeseen adverse events.
- In addition, the positive currency impact on 2015 full-year business EPS is estimated to be approximately +12%, under the assumption that exchange rates remain stable in the following three quarters at the average rates of March 2015.
To access the full press release of the Q1 2015 results, please click here.
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These statements include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects”, “anticipates”, “believes”, “intends”, “estimates”, “plans” and similar expressions. Although Sanofi’s management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include among other things, the uncertainties inherent in research and development, future clinical data and analysis, including post marketing, decisions by regulatory authorities, such as the FDA or the EMA, regarding whether and when to approve any drug, device or biological application that may be filed for any such product candidates as well as their decisions regarding labelling and other matters that could affect the availability or commercial potential of such product candidates, the absence of guarantee that the product candidates if approved will be commercially successful, the future approval and commercial success of therapeutic alternatives, the Group’s ability to benefit from external growth opportunities, trends in exchange rates and prevailing interest rates, the impact of cost containment policies and subsequent changes thereto, the average number of shares outstanding as well as those discussed or identified in the public filings with the SEC and the AMF made by Sanofi, including those listed under “Risk Factors” and “Cautionary Statement Regarding Forward-Looking Statements” in Sanofi’s annual report on Form 20-F for the year ended December 31, 2014. Other than as required by applicable law, Sanofi does not undertake any obligation to update or revise any forward-looking information or statements.