| Media Relations: | Investor Relations: |
|---|---|
| Anna Robinson | George Grofik |
| 908-989-0726 | 908-981-5560 |
| Email: [email protected] | Email: [email protected] |





Solid Performance in the First Quarter of 2016 with Business EPS up 5.3% at Constant Exchange Rates
Paris, April 29, 2016 / PRNewswire / — Sanofi (NYSE: SNY; EURONEXT: SAN)
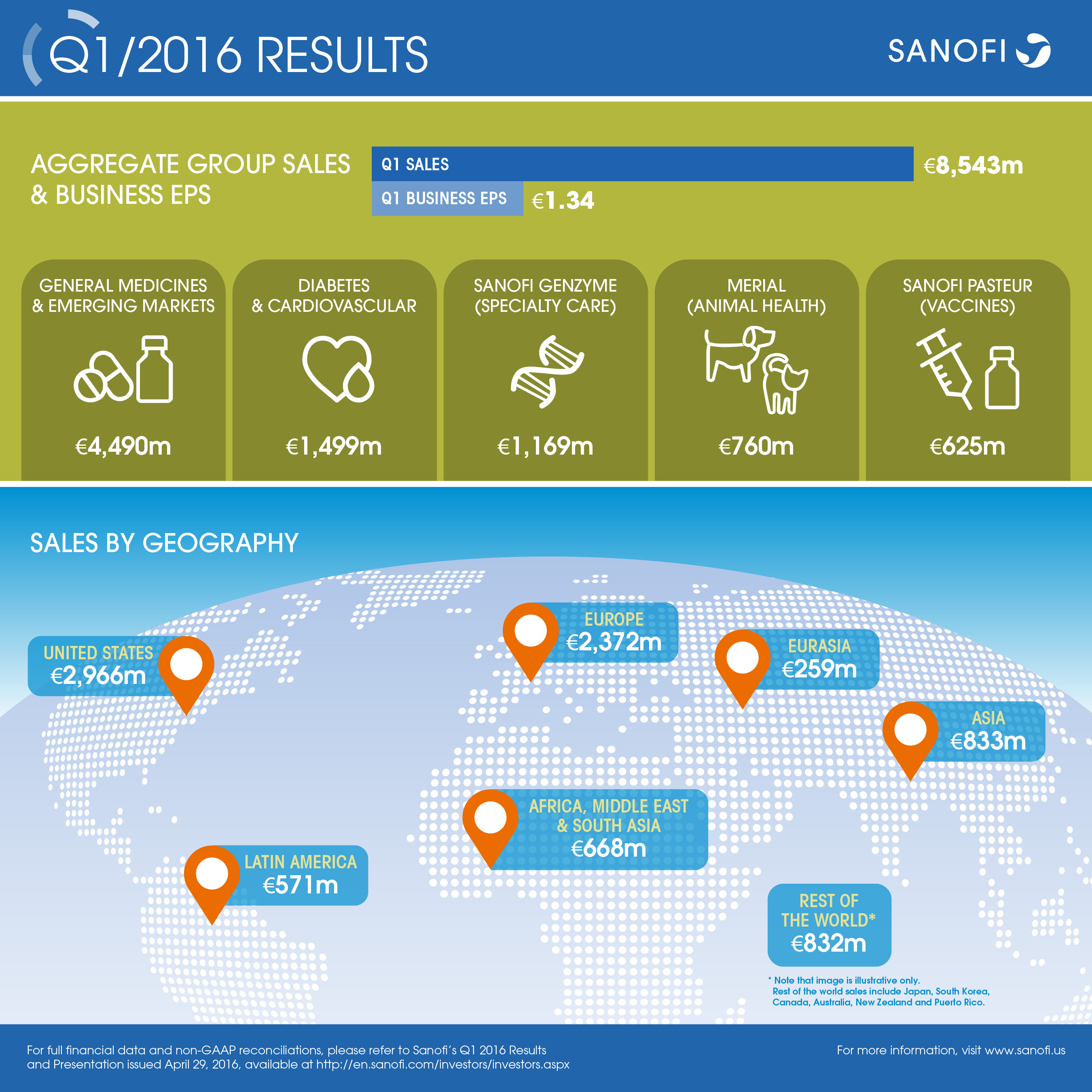
| Q4 2016 | Change | Change (CER) | |
| Aggregate Group sales(1) | €8,543m | -1.9% | +0.7% |
| Business net income(2) | €1,722m | -0.2% | +3.5% |
| Business EPS(3) | €1.34 | +1.5% | -12.9% |
(1) Including Animal Health Business (see Appendix 8 for definition of Aggregate Group sales) which is reported on a single line in the consolidated income statements in accordance with IFRS 5 (Non-current assets held for sale and discontinued operations). Additionally, Sanofi comments include Animal Health Business for every income statement line using “Aggregate” wording. (2) In order to facilitate an understanding of operational performance, Sanofi comments on the business net income statement. Business net income is a non-GAAP financial measure (see Appendix 8 for definition). The consolidated income statement for Q1 2016 is provided in Appendix 4 and a reconciliation of business net income to IFRS net income reported in Appendix 3; (3) (EPS) Earnings Per Share.
Sanofi Chief Executive Officer, Olivier Brandicourt, commented:
“I am pleased with the solid performance of the Group in the first quarter driven by Sanofi Genzyme, Sanofi Pasteur and Merial as well as our growth in Emerging Markets. At the same time, we have made significant progress with two major late-stage pipeline assets, dupilumab and sarilumab, highlighting the potential of our emerging immunology franchise. As we enter the second quarter, we remain focused on the execution of our strategic priorities and confirm our financial outlook of broadly stable Business EPS at CER for the full year.”
Solid financial results and 2016 Guidance confirmed
- Aggregate Group sales increased by 0.7% (-1.9% at 2016 exchange rates) to €8,543 million (with VaxServe sales of non-Group products of €83 million now reported in Other revenues). Excluding Venezuela, Aggregate Group sales grew 3.0%
- Business EPS was up 5.3% at CER to €1.34 and increased 1.5% on a reported basis
- Sanofi continues to expect 2016 Business EPS to be broadly stable at CER, barring unforeseen major adverse events
Sales performance led by Sanofi Genzyme and Emerging Markets
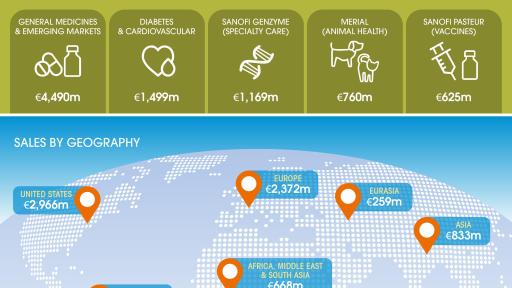
- Sanofi Genzyme Global Business Unit (GBU) sales increased 20.5%, driven by the MS franchise
- Strong growth of Sanofi Pasteur GBU with sales of €625m, up +8.2%, despite expected lower sales of Pentacel® due to supply constraints in the U.S.
- Diabetes and Cardiovascular GBU performance reached €1,499 million, down 5.8%. On a worldwide basis, including Emerging Markets, Diabetes franchise sales declined 4.5%
- General Medicines & Emerging Markets GBU sales of €4,490 million, -4.3%, or stable excluding Venezuela
- Total Emerging Markets sales were €2,373 million, an increase of 13.1% excluding Venezuela
Advancing the next wave of innovation
- Dupilumab is the first systemic therapy to show positive Phase III results in moderate-to-severe atopic dermatitis, representing a promising new class of immunotherapies
- Sarilumab demonstrated superiority vs. adalimumab in a Phase III monotherapy study in rheumatoid arthritis
- The WHO Strategic Advisory Group of Experts on Immunization recommended the use of Dengvaxia® in endemic countries. The first public dengue immunization program started in the Philippines in April
R&D Update
Regulatory update
Regulatory updates since the publication of the fourth quarter results on February 9, 2016 include the following:
- In February, the U.S. Food and Drug Administration (FDA) accepted the New Drug Application (NDA) for the investigational fixed-ratio combination of basal insulin glargine and GLP-1 receptor agonist lixisenatide for the treatment of adults with type 2 diabetes. The FDA decision is anticipated in August 2016. In March, the FDA announced that on May 25, 2016, the Endocrinologic and Metabolic Drugs Advisory Committee will review the NDA for the investigational fixed-ratio combination of basal insulin glargine and lixisenatide and investigational lixisenatide. In Europe, Sanofi submitted the dossier of this fixed-ratio combination to health authorities in March.
- In February, the new hexavalent pediatric vaccine, PR5i (DTP-HepB-Polio-Hib), was granted a Marketing Authorization in EU.
At the end of April 2016, the R&D pipeline contained 46 pharmaceutical new molecular entities (excluding Life Cycle Management) and vaccine candidates in clinical development of which 15 are in Phase III or have been submitted to the regulatory authorities for approval.
Portfolio update
Phase III:
- The Data Monitoring Committee (DMC) of the ODYSSEY OUTCOMES study for Praluent® has completed the first interim analysis based on unblinded study data. In addition to the review of the safety data, the DMC performed a futility assessment and recommended the study continue with no changes. Sanofi remains blinded to the actual results of this analysis. The second interim analysis for futility and overwhelming efficacy potentially could occur in the second half of 2016 when 75% of the targeted number of primary events have occurred.
- In March, Sanofi and Regeneron announced positive topline results from the Phase III ODYSSEY ESCAPE trial evaluating Praluent® (alirocumab) Injection in patients with an inherited form of high cholesterol known as heterozygous familial hypercholesterolemia, whose cholesterol levels required chronic, weekly or bi-weekly apheresis therapy.
- In March, Sanofi and Regeneron announced that a Phase III monotherapy study, SARIL-RA-MONARCH, met its primary endpoint demonstrating that sarilumab was superior to adalimumab (marketed by AbbVie as Humira®) in improving signs and symptoms in patients with active rheumatoid arthritis at Week 24.
- In April, Sanofi and Regeneron announced positive topline results from two placebo-controlled Phase 3 studies known as LIBERTY AD SOLO 1 and SOLO 2, evaluating investigational dupilumab in adult patients with inadequately controlled moderate-to-severe atopic dermatitis. In the studies, treatment with dupilumab as monotherapy significantly improved measures of overall disease severity, skin clearing, itching, quality of life, and mental health.
- The second generation meningococcal ACYW conjugate vaccine, Men Quad TT, with a broader age indication (from infants to the elderly) entered into Phase III.
Phase II:
- In March, data from a Phase I/II clinical study, NEO1, evaluating the investigational novel enzyme replacement therapy neoGAA in 24 patients with late-onset Pompe disease were presented at WORLD Symposium 2016. The safety and efficacy data from this study support further development of the therapy. Sanofi plans to begin enrolling patients in a pivotal Phase 3 trial for neoGAA in Q2 2016.
- SAR422459, ABC4A gene therapy, entered in Phase IIa in Stargardt Disease, a rare eye disease.
To access the full press release of the Q1 2016 earnings results, please click here.
About Sanofi
Sanofi, a global healthcare leader, discovers, develops and distributes therapeutic solutions focused on patients' needs. Sanofi has core strengths in diabetes solutions, human vaccines, innovative drugs, consumer healthcare, emerging markets, animal health and Genzyme. Sanofi is listed in Paris (EURONEXT: SAN) and in New York (NYSE: SNY).
Sanofi is the holding company of a consolidated group of subsidiaries and operates in the United States as Sanofi US. For more information on Sanofi US, please visit https://www.sanofi.us and https://www.news.sanofi.us/social-media or call 1-800-981-2491.
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These statements include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects”, “anticipates”, “believes”, “intends”, “estimates”, “plans” and similar expressions. Although Sanofi’s management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include among other things, the uncertainties inherent in research and development, future clinical data and analysis, including post marketing, decisions by regulatory authorities, such as the FDA or the EMA, regarding whether and when to approve any drug, device or biological application that may be filed for any such product candidates as well as their decisions regarding labelling and other matters that could affect the availability or commercial potential of such product candidates, the absence of guarantee that the product candidates if approved will be commercially successful, the future approval and commercial success of therapeutic alternatives, the Group’s ability to benefit from external growth opportunities, trends in exchange rates and prevailing interest rates, the impact of cost containment initiatives and subsequent changes thereto, the average number of shares outstanding as well as those discussed or identified in the public filings with the SEC and the AMF made by Sanofi, including those listed under “Risk Factors” and “Cautionary Statement Regarding Forward-Looking Statements” in Sanofi’s annual report on Form 20-F for the year ended December 31, 2015. Other than as required by applicable law, Sanofi does not undertake any obligation to update or revise any forward-looking information or statements.