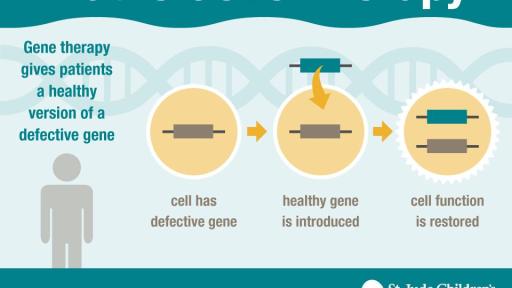
Gene therapy pioneered at St. Jude shows early success against “Bubble Boy” disease
Early results suggest novel gene therapy is safe and effective for treatment of inherited immune disease, according to St. Jude Children’s Research Hospital and the National Institute of Allergy and Infectious Diseases
PR Newswire – (MEMPHIS, Tenn. – April 20, 2016)
Adolescents and young adults with a severe inherited immunodeficiency disorder improved following treatment with novel gene therapy developed at St. Jude Children’s Research Hospital and at the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health. The results of this study appear today in the journal Science Translational Medicine.
The study involved five males with X-linked severe combined immunodeficiency disease (SCID-X1), also known as “Bubble Boy” disease, who were all treated at NIAID. This inherited disorder involves a mutation in the IL2RG gene that affects males and occurs in 1 of every 50,000 to 100,000 live births, leaving them with little to no immune protection.
Health & immune function improve #SCID patients following #genetherapy with novel vector. http://bit.ly/1VC5yzN Tweet
“This study demonstrates that lentivirus gene therapy, when combined with busulfan conditioning, can rebuild the immune system and lead to broad immunity in young adults with this devastating disorder,” said co-senior author Brian Sorrentino, M.D., a member of the St. Jude Department of Hematology. The vector, a re-engineered lentivirus that is used to deliver a correct copy of the mutant gene to the blood-producing stem cells of patients, was developed and produced at St. Jude.
“While additional clinical experience and follow-up is needed, these promising results suggest gene therapy should be considered as an early treatment for patients in order to minimize or prevent the life-threatening organ damage that occurs when bone marrow transplant therapy fails to provide a sufficient immune response,” Sorrentino said. The study’s first and corresponding author is Suk See De Ravin, M.D., Ph.D., of NIAID. The co-corresponding author is Harry Malech, M.D., of NIAID.
Patients in this safety and efficacy study were 7 to 23 years old. They all had chronic viral infections and other health problems related to SCID-X1 after one or more bone marrow transplants failed to fully correct their immune function. Like two-thirds of SCID-X1 patients, study participants lacked a tissue-matched sibling to serve as a hematopoietic stem cell donor. Instead, they had received blood-forming stem cells without any chemotherapy or myeloid conditioning from a parent donor who provided only a partial genetic match.
The clinical trial included a number of firsts for gene therapy of SCID-X1, including the first use of a lentiviral vector and a streamlined stable production system that increased the vector supply. The vector included novel features designed to increase safety and effectiveness. The study also marked the first use of the chemotherapy drug busulfan in gene therapy for SCID-X1 to help the gene-corrected stem cells become re-established and functional after being infused back into SCID-X1 patients.
More than two years after undergoing gene therapy, the first two patients were producing a greater percentage of immune cells, including T, B and natural killer cells, with the corrected gene. The gene therapy had also restored antibody production in response to vaccination. The percentage of immune cells with the corrected gene ranged from 13 to 55 percent of T cells to 38 percent of B cells and 56 to 76 percent of NK cells, and 9 to 10 percent of bone marrow stem cells. This high level of genetic correction in all different types of blood cells was not obtained in previous gene therapy studies for SCID-X1.
The patients’ health also improved as their chronic viral infections resolved and they put on weight as their protein absorption improved. One patient’s disfiguring warts also eased. Both ended life-long immune globulin therapy. Despite the improvements, one patient died from pre-existing lung damage more than two years after receiving gene therapy, which highlights the importance of earlier intervention before irreversible organ damage occurs.
In addition, promising levels of the normal gene from the gene therapy increasing in multiple immune cell types were reported in the three other study patients six to nine months after gene therapy.
Safety results were also reassuring. There was also no indication of possible pre-cancer cell proliferation. “The lentiviral vector has design features, including genetic insulators to block activation of adjacent genes, that make it fundamentally safer,” Sorrentino said.
Meanwhile, St. Jude has opened a gene therapy trial using the same lentiviral vector and busulfan conditioning for newly identified infants with SCID-X1 who lack a genetically matched sibling hematopoietic stem cell donor. “Based on the safety and health benefits for older patients reported in this study, we hope this novel gene therapy will help improve immune functioning and transform the lives of younger patients with this devastating disease,” said Sorrentino, who is leading this study with Ewelina Mamcarz, M.D., an assistant member of the St. Jude Department of Bone Marrow Transplantation and Cell Therapy.
The other authors of the current study are R. E. Throm, S. Zhou and M. Meagher, all of St. Jude; X. Wu, L. Su and D. Kuhns, all of Frederick National Laboratory for Cancer Research, Frederick, MD; S. Moir, L. Kardava, S. Anaya O’Brien, J. Ulrick, N. Kwatemaa, P. Littel, N. Theobald, U. Choi, M. Marquesen, D. Hilligoss, J. Lee, C.M. Buckner, K.A. Zarember, E. Kang and C. Hadigan, all of NIAID; G. O’Connor and D. McVicar, both of the National Cancer Institute; L.D. Notarangelo, Boston Children’s Hospital and Harvard Medical School; I.C. Hanson, Texas Children’s Hospital, Houston; M.J. Cowan, Benioff Children’s Hospital and University of California, San Francisco; and J.T. Gray, Audentes Therapeutics, San Francisco.
The study was funded in part by grants (A100644, A100988, HL53749) from the NIH; the Assisi Foundation of Memphis, and ALSAC.
St. Jude Media Relations Contacts
Jann Ingmire
Desk: (901) 595-6384
Cell: (901) 356-5716
[email protected]
[email protected]
Frannie Marmorstein
Desk: (901) 595-0221
Cell: (901) 379-6072
[email protected]
[email protected]