Allergan Breaks Ground on $200 Million Waco Facility Expansion
-- Expansion to add more than 100 full time jobs to local Waco and Central Texas economies --
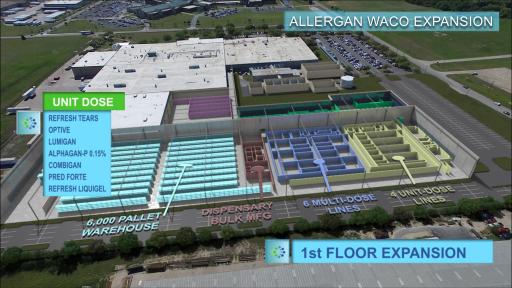
-- Will add 322,000 square feet of manufacturing space, nearly doubling current footprint –
-- Will increase Waco facility production capacity by more than 50 percent --
-- Provides manufacturing capabilities for more than 40 different product formulations –
-- Expansion expected to be completed by 2020 --
-- Baylor University analysis shows Allergan expansion to add $380 million in economic impact during construction, $460 million in ongoing impact to Central Texas economy -
PR Newswire, WACO, TX – April 25, 2016
Allergan plc (NYSE: AGN), a leading global pharmaceutical company, today broke ground on a $200 million expansion of its Waco, Texas facility. The expansion will add 322,000 square feet of manufacturing space to the current facility, nearly doubling its current footprint. Once completed, the expansion is expected to add approximately 100 full-time jobs, with the potential for the expanded operations adding as many as 250 full-time jobs as the facility reaches full utilization. New positions that will be added include chemists, microbiologists, process engineers and production and maintenance technicians.
Additionally, the facility expansion will upgrade the Company’s manufacturing capabilities and capacity. The expansion will add a new raw material dispensary, an automated bulk formulation suite and ten new state-of-the-art production lines as well as warehousing space. Upon completion, the Waco facility will be capable of producing more than 40 different product formulations and will increase its production capacity by more than 50 percent.
The construction, commissioning and validation of the facility expansion and production processes are expected to be completed by 2020. Allergan’s Waco facility is the Company’s flagship manufacturing facility for its leading eye care products, including Restasis®, Lumigan®, Combigan®, Refresh Plus®, Refresh Tears® and Latisse®.
“Today’s groundbreaking marks an important milestone for Allergan, strengthening our commitment to our people, operations and the partnership we have built with Waco and the Central Texas communities for 27 years,” said Brent Saunders, President and CEO of Allergan. “This investment will add highly-skilled jobs to the Waco and Central Texas economies, elevate our manufacturing capabilities and increase our ability to provide more complex medicines to more physicians and their patients around the globe.”
“As we celebrate this expansion today, it is important to remember that it was achieved through great collaboration between Allergan and the local Waco and Texas state governments and chambers who have been great partners in providing a positive business climate for our success. It is through these open collaborations that companies like Allergan can continue to succeed in providing important products, needed innovation and highly skilled jobs for Texas and the U.S. economy,” added Saunders.
"I applaud Allergan for their continued commitment to Waco, and this expansion which will create important new jobs for highly skilled Texas workers in our growing life sciences industry,” said Texas Lieutenant Governor Dan Patrick. “I am confident that Texas’ extraordinary business climate and our commitment to growing industries like the life sciences will continue to attract healthcare industry leaders like Allergan to the Lone Star State."
AGN breaks ground on $200 million expansion of Waco, TX Facility Tweet
“As a member of Congress, I have worked to ensure that our federal laws and policies equip the private sector with the resources and flexibility necessary to be at the forefront of medical innovation for individuals living with serious illnesses and conditions,” said U.S. Congressman Michael C. Burgess, M.D. (R-TX). “As a country, we must maintain our commitment to creating a positive business environment that attracts the best and brightest to invest in innovation—this requires providing a competitive corporate tax policy, transparent and consistent regulation, and a strong and skilled workforce able to help produce complex medical products. If we are successful in doing these things, we can strengthen our economy and improve the lives of all Americans. I am pleased that Allergan has decided to invest its best and brightest to advance innovation in Texas.”
In addition to today’s groundbreaking, Tom Kelly, Ph.D., Baylor Center for Business & Economic Research in Baylor’s Hankamer School of Business provided a supplement to his previous economic impact analysis, “The Economic Impact of Allergan on the Central Texas Economy,” to capture the impact of the expansion. Dr. Kelly’s economic impact analysis supplement1 found:
- Including multiplier effects, the expansion will add $380 million to the Central Texas economy during the construction period.
- During the first year of construction the total economic impact of Allergan capital investment plus operations on the Central Texas economy is projected to increase to $522.3 million from a previously projected $450.3 million in 2016.
- During the remainder of the construction period the average annual economic impact of new capital investment will increase to $72 million per year.
- Upon completion of the capital expansion, Allergan’s annual economic impact on the Central Texas economy will be $461.3 million compared with the previously projected $407.2 million per year.
"This supplement continues to underscore the clear economic and job-creating value Allergan has and will continue to provide to both the Waco and Central Texas economies. We hope that Allergan serves as a benchmark for other companies looking for a business friendly community with a highly skilled workforce to consider relocating operations to Waco and the Central Texas area," said Tom Kelly, Ph.D., Baylor Center for Business & Economic Research.
Full-text of the original report and the supplement is available via the following links:
January 2016 Economic Impact Study: https://allergan-web-cdn-prod.azureedge.net/actavis/actavis/media/allergan-pdf-documents/pdf-documents/allergan-baylor-university-study-jan-2016_1.pdf
April 2016 Economic Impact Study Supplement: https://allergan-web-cdn-prod.azureedge.net/actavis/actavis/media/allergan-pdf-documents/pdf-documents/allergan-baylor-study-update_expansion_4-2016.pdf
About Allergan’s Waco, TX Operations
Allergan employs more than 750 highly skilled workers at its 400,000 square-foot facility located on a 69-acre campus in Waco’s Texas Central Park. The facility currently produces a number of eye-care and dermatological products, including Restasis® (Cyclosporine Ophthalmic Emulsion) 0.05%, a treatment that helps increase the eyes’ natural ability to produce tears, which may be reduced by inflammation due to Chronic Dry Eye, and Latisse® (bimatoprost ophthalmic solution) 0.03% an FDA-approved treatment to grow eyelashes for people with inadequate or not enough lashes. Allergan opened its Waco facility in 1989, and in 2016, the Company began a substantial expansion of the facility to add a new testing laboratory and additional production capacity to meet the growing demand for its products around the world.
About Allergan
Allergan plc (NYSE: AGN), headquartered in Dublin, Ireland, is a unique, global pharmaceutical company and a leader in a new industry model—Growth Pharma. Allergan is focused on developing, manufacturing, and commercializing innovative branded pharmaceuticals, high-quality generic and over-the-counter medicines, and biologic products for patients around the world.
Allergan markets a portfolio of best-in-class products that provide valuable treatments for the central nervous system, eye care, medical aesthetics, gastroenterology, women's health, urology, cardiovascular and anti-infective therapeutic categories, and operates the world's third-largest global generics business, providing patients around the globe with increased access to affordable, high-quality medicines. Allergan is an industry leader in research and development, with one of the broadest development pipelines in the pharmaceutical industry and a leading position in the submission of generic product applications globally.
With commercial operations in approximately 100 countries, Allergan is committed to working with physicians, health care providers, and patients to deliver innovative and meaningful treatments that help people around the world live longer, healthier lives.
For more information, visit Allergan's website at www.allergan.com.
Forward-Looking Statement
Statements contained in this press release that refer to future events or other non-historical facts are forward-looking statements that reflect Allergan's current perspective of existing trends and information as of the date of this release. Except as expressly required by law, Allergan disclaims any intent or obligation to update these forward-looking statements. Actual results may differ materially from Allergan's current expectations depending upon a number of factors affecting Allergan's business. These factors include, among others, the difficulty of predicting the timing or outcome of FDA approvals or actions, if any; the impact of competitive products and pricing; market acceptance of and continued demand for Allergan's products; difficulties or delays in manufacturing; and other risks and uncertainties detailed in Allergan's periodic public filings with the Securities and Exchange Commission, including but not limited to Allergan's Annual Report on Form 10-K for the year ended December 31, 2015 (certain of such periodic public filings having been filed under the "Actavis plc" name). Except as expressly required by law, Allergan disclaims any intent or obligation to update these forward-looking statements.
###
1Supplementary analysis results based on initial estimate of 100 additional jobs added post-expansion.
Links
Allergan.comResources
Baylor University Economic Impact Study – January 2016 Baylor University Economic Impact Study Supplement – April 2016
Media:
Mark Marmur
(862) 261-7558