Boston Scientific receives FDA clearance for the Asurys™ Fluid Management System
New technology designed to provide a single irrigation management system for ureteroscopy, cystoscopy, percutaneous nephrolithotomy (PCNL) and benign prostatic hyperplasia (BPH) procedures
MARLBOROUGH, Mass., March 30, 2026 – Boston Scientific Corporation (NYSE: BSX) today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for the Asurys™ Fluid Management System, designed to provide irrigation and distention during endoscopic urologic procedures, including ureteroscopies.
Ureteroscopy is a common procedure to diagnose and treat problems in the urinary tract, most commonly kidney stones.1 Research shows that about one in 10 people will develop a kidney stone at some point in their lives.2 One challenge during ureteroscopy is maintaining fluid irrigation flow for clear visualization while keeping intrarenal pressure (IRP) low to help promote safe patient care.3-5 Elevated IRP may contribute to a range of post-operative complications including systemic inflammatory response syndrome (SIRS), sepsis and renal damage.6-10
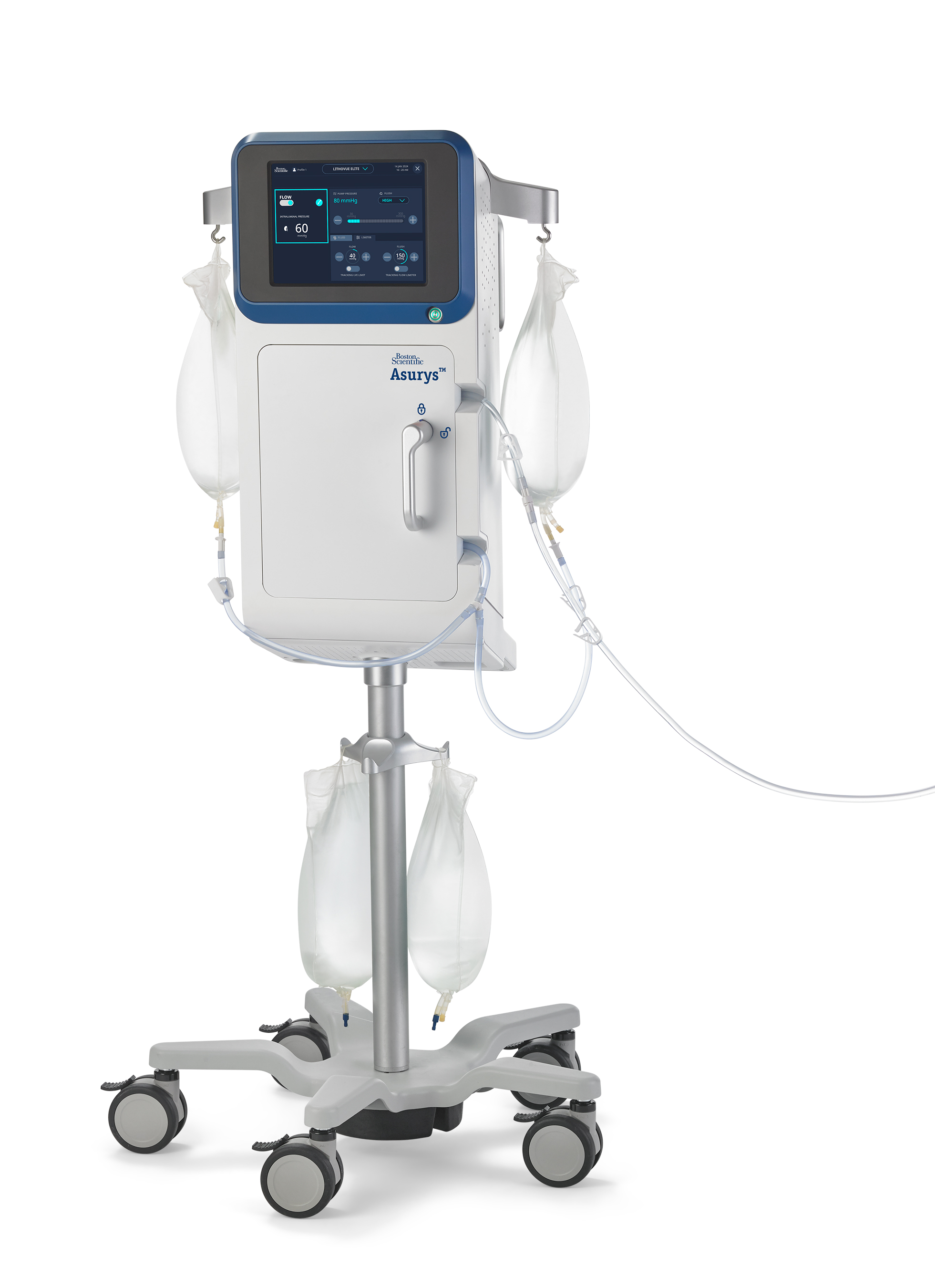
The Asurys System is the latest device in Boston Scientific’s StoneSmart™ Solutions ecosystem of products to manage and treat kidney stones. When paired with the LithoVue™ Elite Single-Use Digital Flexible Ureteroscope with IRP monitoring, the Asurys System is designed to aid in procedural IRP management.11
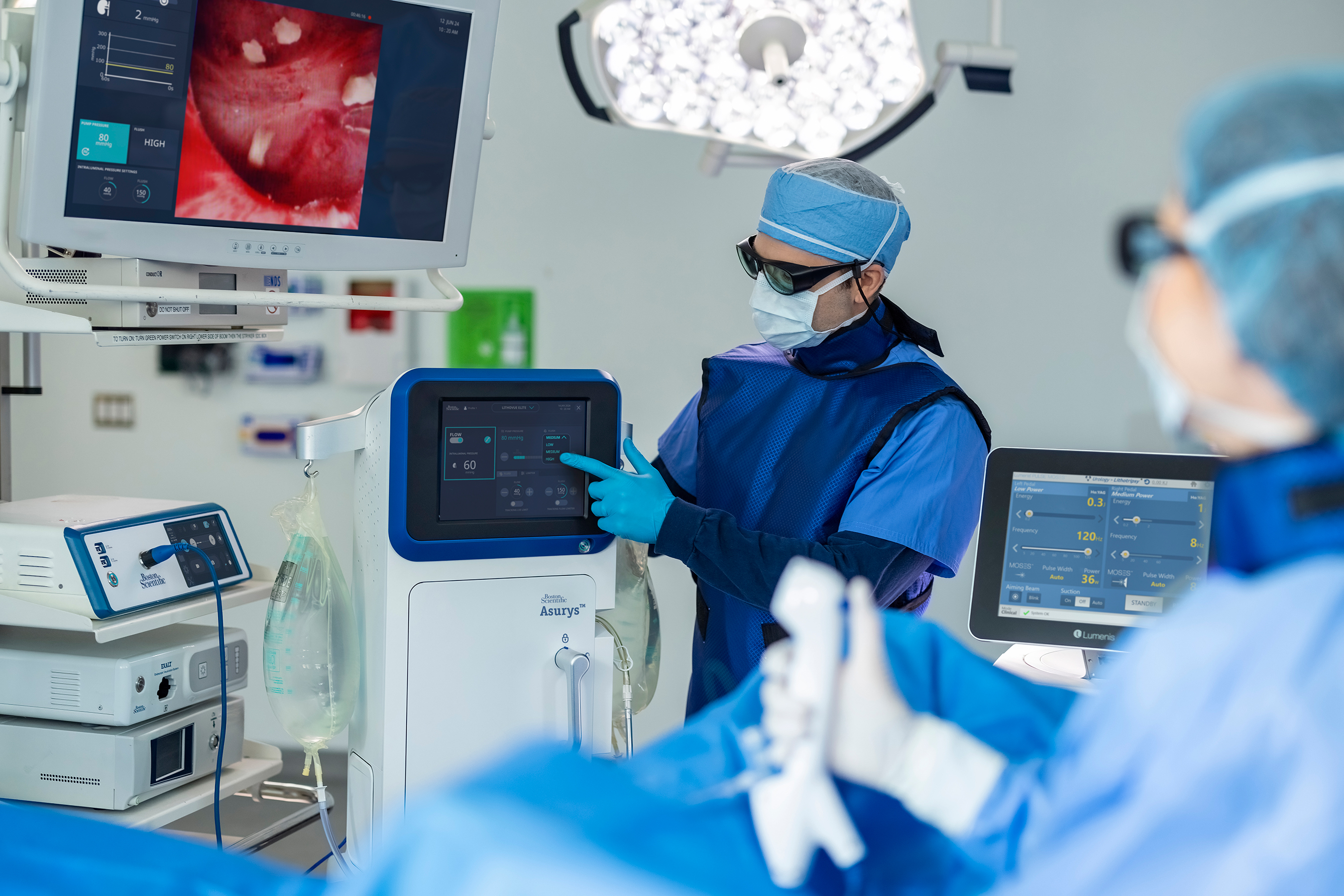
“Based on feedback gathered through our work with urologists, we know measuring and managing intrarenal pressure (IRP) while maintaining irrigation flow for visualization is increasingly important,” said Dr. Ronald Morton, chief medical officer, Urology, Boston Scientific. “Until now, there has not been a system that can automatically respond to IRP data in real-time. The Asurys Fluid Management System can automatically regulate irrigation inflow to help minimize the potential of exceeding physician-selected IRP settings.”
Another integrated feature of the Asurys System, when connected to the LithoVue Elite System, includes irrigation control from the scope handle, which is designed to reduce reliance on support staff. By pressing a button on the LithoVue Elite Ureteroscope, physicians can activate the on-demand flush feature to reposition stones into a better location for lithotripsy or basketing without manual irrigation.
“We’re driven to advance urology for the long term by making meaningful improvements to patient outcomes while developing innovative technologies that equip and support care teams,” said Meghan Scanlon, senior vice president and president, Urology, Boston Scientific. “The Asurys Fluid Management System is a clinical solution intended to streamline workflows and ultimately improve the treatment of kidney stone disease, all with the aim of reducing the cognitive burden for endourologists.”
In addition to ureteroscopies, the Asurys System can be used as an irrigation management option for cystoscopy, percutaneous nephrolithotomy (PCNL) and benign prostatic hyperplasia (BPH) procedures. Limited market release of the device in the U.S. will begin in the coming days.
For more information, please visit www.bostonscientific.com/Asurys or www.bostonscientific.com/StoneSmart.
About Boston Scientific
Boston Scientific transforms lives through innovative medical technologies that improve the health of patients around the world. As a global medical technology leader for more than 45 years, we advance science for life by providing a broad range of high-performance solutions that address unmet patient needs and reduce the cost of healthcare. Our portfolio of devices and therapies helps physicians diagnose and treat complex cardiovascular, respiratory, digestive, oncological, neurological and urological diseases and conditions. Learn more at www.bostonscientific.com and follow us on LinkedIn.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements may be identified by words like "anticipate," "expect," "project," "believe," "plan," "estimate," "intend" and similar words. These forward-looking statements are based on our beliefs, assumptions and estimates using information available to us at the time and are not intended to be guarantees of future events or performance. These forward-looking statements include, among other things, statements regarding our business plans and product performance and impact, clinical trials, and new and anticipated product approvals and launches. If our underlying assumptions turn out to be incorrect, or if certain risks or uncertainties materialize, actual results could vary materially from the expectations and projections expressed or implied by our forward-looking statements. These factors, in some cases, have affected and in the future (together with other factors) could affect our ability to implement our business strategy and may cause actual results to differ materially from those contemplated by the statements expressed in this press release. As a result, readers are cautioned not to place undue reliance on any of our forward-looking statements.
Factors that may cause such differences include, among other things: future economic, competitive, reimbursement and regulatory conditions; geopolitical events; manufacturing, distribution and supply chain disruptions and cost increases; disruptions caused by cybersecurity events; disruptions caused by public health emergencies or extreme weather or other climate change-related events; labor shortages and increases in labor costs; variations in outcomes of ongoing and future clinical trials and market studies; new product introductions; expected procedural volumes; the closing and integration of acquisitions; demographic trends; intellectual property; litigation; financial market conditions; and future business decisions made by us and our competitors. All of these factors are difficult or impossible to predict accurately and many of them are beyond our control. For a further list and description of these and other important risks and uncertainties that may affect our future operations, see Part I, Item 1A – Risk Factors in our most recent Annual Report on Form 10-K filed with the Securities and Exchange Commission, which we may update in Part II, Item 1A – Risk Factors in Quarterly Reports on Form 10-Q we have filed or will file hereafter. We disclaim any intention or obligation to publicly update or revise any forward-looking statements to reflect any change in our expectations or in events, conditions or circumstances on which those expectations may be based, or that may affect the likelihood that actual results will differ from those contained in the forward-looking statements, except as required by law. This cautionary statement is applicable to all forward-looking statements contained in this document.
CONTACTS:
Nate Gilbraith
Media Relations
+1 (701) 212-9589
[email protected]
Lauren Tengler
Investor Relations
+1 (508) 683-4479
[email protected]
References:
- National Institute of Diabetes and Digestive and Kidney Diseases. Cystoscopy & Ureteroscopy. Available at: https://www.niddk.nih.gov/health-information/diagnostic-tests/cystoscopy-ureteroscopy. Accessed July 2025.
- National Kidney Foundation. Kidney Stones. Available at: https://www.kidney.org/atoz/content/kidneystones. Accessed July 2025.
- Yuen SKK, Zhong W, Chan YS, et al. Current utility, instruments, and future directions for intra-renal pressure management during ureteroscopy: Scoping review by global research in intra-renal pressure collaborative group initiative. Ther Adv Urol. 2025;17.
- Somani B, Davis N, Emiliani E, et al. Intrarenal pressure monitoring during ureteroscopy: A Delphi panel consensus. Eur Urol Open Sci. 2025;73:43–50.
- Williams JG, Rouse L, Turney BW, Waters SL, Moulton DE. A lumped-parameter model for kidney pressure during stone removal. IMA J Appl Math. 2020;85:703–723.
- Tokas T, Herrmann TRW, Skolarikos A, et al. Pressure matters: intrarenal pressures during normal and pathological conditions, and impact of increased values to renal physiology. World J Urol. 2019 Jan;37(1):125-31.
- Gutierrez-Aceves J, Negrete-Pulido O, Avila-Herrera P. Preoperative Antibiotics and Prevention of Sepsis in Genitourinary Surgery. In Smith AD, Badlani GH, Preminger GM, Kavoussi LR (Eds.), Smith’s Textbook of Endourology. New York, NY: Blackwell Publishing Ltd. 2012:38–52.
- Osther PJS, Pedersen KV, Lildal SK, Pless MS, Andreassen KH, Osther SS, Jung HU. Pathophysiological aspects of ureterorenoscopic management of upper urinary tract calculi. Curr Opin Urol.2016;26:63–69
- Schwalb DM, Eshghi M, Davidian M, et al. Morphological and physiological changes in the urinary tract associated with ureteral dilation and ureteropyeloscopy: an experimental study. J Urol. 1993 Jun;149(6):1576–85.
- Zhong W, Leto G, Wang L, Zeng G. Systemic inflammatory response syndrome after flexible ureteroscopic lithotripsy: a study of risk factors. J Endourol. 2015 Jan;29(1):25-8.
- Morris, A. S., & Langari, R. (2021). Intelligent sensors. Measurement and Instrumentation, 323–348. https://doi.org/10.1016/b978-0-12-817141-7.00011-6