| Media Relations: | Investor Relations: |
|---|---|
| Ashleigh Koss | George Grofik |
| 908-981-8745 | +33 (0)1 53 77 45 45 |
| Email: [email protected] | Email: [email protected] |






Media Panel
Resources
Sanofi delivers 2018 business EPS growth of 5.1% at CER
Paris, February 7, 2019 — Sanofi (NASDAQ: SNY; EURONEXT: SAN)
| Q4 2018 | Change | Change at CER | 2018 | Change | Change at CER |
|
| IFRS net sales reported | €8,997m | +3.5% | +3.9% | €34,463m | -1.7% | +2.5% |
| IFRS net income reported | €254m | +101.6% | - | €4,306m | -48.8%(2) | - |
| IFRS EPS reported | €0.20 | +100.0% | - | €3.45 | -48.5%(2) | - |
| Business net income(1) | €1,364m | +2.9% | +4.3% | €6,819m | -1.8% | +4.2% |
| Business EPS(1) | €1.10 | +3.8% | +4.7% | €5.47 | -0.9% | +5.1% |
Fourth-quarter sales(3) growth driven by Specialty Care and Vaccines
- Net sales were €8,997 million, an increase of 3.5% on a reported basis, 3.9%(3) at CER and 2.6% at CER/CS (4).
- Sanofi Genzyme sales were up 37.4% (16.1% at CER/CS(4)), led by Immunology and Rare Blood Disorder franchises.
- Vaccines sales increased 9.7%, driven by successful influenza differentiation strategy and Menactra®.
- CHC sales increased 1.9%, supported by Emerging Markets.
- DCV(5) GBU sales were down 11.3%; Global Diabetes franchise sales declined 10.5% in line with 2015-2018 guidance.
- Emerging Markets sales(6) were up 6.0%, reflecting strong performance in Asia.
Full-Year 2018 sales growth from new products and Emerging markets more than offset impact of U.S. LoEs
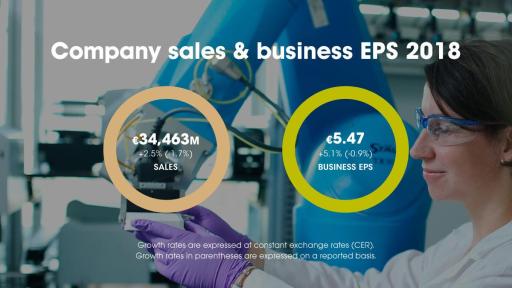
- Net sales in 2018 were €34,463 million, down 1.7% on a reported basis and grew 2.5% at CER (up 0.6% at CER/CS(4)).
- Sanofi Genzyme grew 30.8% (+14.2% at CER/CS(4)) to €7,226 million.
- Vaccines sales increased 2.4% to €5,118 million while CHC sales were up 3.0% to €4,660 million.
- DCV(5) GBU sales declined 13.8% to €4,511 million.
- Emerging Markets sales were up 7.5%, supported by strong performance in China (up 12.7%).
Sanofi delivers 2018 business EPS at the high end of its guidance range
- Q4 2018 business EPS(1) up 4.7% at CER to €1.10.
- Full-Year 2018 business EPS of €5.47 up 5.1% at CER and IFRS EPS of €3.45 (down 48.5%(2)).
- Board proposes dividend of €3.07, the 25th consecutive increase in dividend.
Key achievements in sustaining innovation in R&D
- Isatuximab met primary endpoint of ICARIA phase 3 study in Relapsed/Refractory Multiple Myeloma.
- BIVV001 demonstrated sustained high factor levels at once-weekly dosing with data presented at ASH.
- FDA Priority Review granted for Dupixent® in adolescents with moderate-to-severe atopic dermatitis.
- R&D strategy evolves towards prioritization of Specialty Care and Vaccines, leveraging technology platforms and data science.
2019 financial outlook
- Sanofi expects 2019 business EPS(1) to grow between 3% and 5%(7) at CER, barring unforeseen major adverse events. Applying average January 2019 exchange rates, the positive currency impact on 2019 business EPS is estimated to be between 1% to 2%.
Sanofi Chief Executive Officer, Olivier Brandicourt, commented:
“In the fourth quarter, we continued the momentum of the previous quarter and we delivered 5% full-year business EPS growth, at the high end of our guidance. In 2018, we executed on important launches including Dupixent®, Libtayo® and Cablivi®, as the headwinds from our U.S. LoEs began to moderate. Additionally, the acquisitions of Bioverativ and Ablynx provided the foundation to build a leading Rare Blood Disorder franchise and to enhance our biologic discovery capabilities. As we enter 2019, our focus remains on delivering our business priorities and transforming Sanofi to address the evolving business dynamics facing our industry.”
(1) In order to facilitate an understanding of operational performance, Sanofi comments on the business net income statement. Business net income is a non-GAAP financial measure (see Appendix 10 for definitions). The consolidated income statement for Q4 2018 is provided in Appendix 3 and a reconciliation of reported IFRS net income to business net income is set forth in Appendix 4; (2) Excluding Animal Health gain on disposal, full-year IFRS net income was up 14.5% and full-year IFRS EPS was up 15.3%; (3) Changes in net sales are expressed at constant exchange rates (CER) unless otherwise indicated (see Appendix 10); (4) Constant Structure: Adjusted for Bioverativ acquisition and divestment of European Generics business; (5) DCV: Diabetes and Cardiovascular; (6) See definition page 8; (7) 2018 business EPS was €5.47.
R&D update
Consult Appendix 6 for full overview of Sanofi’s R&D pipeline
R&D strategy
Sanofi is today providing an update on the evolution of its R&D strategy. Consistent with its ambition to be an industry innovation leader, Sanofi has increased its R&D focus on Specialty Care therapy areas (Oncology, Immunology, Rare Disease and Rare Blood Disorder) while maintaining its commitment to Vaccines. Since 2017, the number of R&D programs in these areas has increased significantly, and they now represent over 90% of Sanofi’s clinical portfolio. This change reflects advances in the Company’s R&D capabilities and understanding of human biology.
In support of this strategy, Sanofi recently carried out a rigorous pipeline prioritization review to accelerate investment behind its most promising programs and to discontinue those with a less attractive expected return profile. As a result, the Company is accelerating the development of 17 programs, including 8 in Oncology. Thirteen development projects and 25 research projects are being discontinued to enhance the company’s focus on delivering first and best in class medicines. Overall, Sanofi could potentially submit 9 new medicines and 25 additional indications to regulatory authorities over 2019 to 2022.
Through the development of its own expertise and the establishment of partnerships with industry pioneers, Sanofi has access to a broad range of therapeutic modalities that enable a more customized, science-driven approach to targeting disease. This includes development of next-generation biologics, such as multi-specific antibodies and Nanobodies, which provide new opportunities relative to traditional monoclonal antibodies in areas such as oncology and immunology, as well as gene therapies. The Company is also employing data science and machine learning across the R&D organization to generate higher quality data, accelerate development and regulatory submissions, and reduce costs. Sanofi expects to maintain an annual R&D budget of approximately €6 billion through 2021.
Regulatory update
Regulatory updates since October 31, 2018 include the following:
- In February, the European Medicine Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval of Praluent® (collaboration with Regeneron) in European Union to reduce cardiovascular risk in people with established atherosclerotic cardiovascular disease.
- In December, the FDA approved the hexavalent vaccine, Vaxelis™, for use in children from 6 weeks through 4 years of age. Vaxelis™ was developed as part of a joint partnership between Sanofi and Merck in the U.S. and Canada. Commercial supply will not be available in the U.S. prior to 2020.
- In December, Dupixent® ( collaboration with Regeneron) was submitted to the FDA for the treatment of adults with inadequately-controlled chronic rhinosinusitis with nasal polyps (CRSwNP).
- In December, the European Commission granted marketing authorization for Dengvaxia® to prevent dengue disease in individuals 9-45 years of age with a documented prior dengue infection and who are living in endemic areas.
- In November, the CHMP recommended approval in European Union of fexinidazole the first all-oral treatment for sleeping sickness.
- In November, the FDA accepted for Priority Review the supplemental Biologics License Application (sBLA) for Dupixent® in adolescent patients 12 to 17 years of age with moderate-to-severe atopic dermatitis, whose disease is inadequately controlled with topical therapies or for whom topical treatment is medically inadvisable. The target action data for the FDA decision is March 11, 2019.
At the beginning of February 2019, the R&D pipeline contained 81 projects including 33 new molecular entities in clinical development. 35 projects are in phase 3 or have been submitted to the regulatory authorities for approval.
Portfolio update
Phase 3:
- In February, Sanofi announced that Isatuximab phase 3 trial (ICARIA study) met primary endpoint of prolonging progression free survival in patients with relapsed/refractory multiple myeloma
- In January, the New England Journal of Medicine (NEJM) published positive results of the Phase 3 trial of Cablivi® (caplacizumab) in adults with acquired thrombotic thrombocytopenic purpura (aTTP).
- In November, new analyses on mortality from the ODYSSEY OUTCOMES trial evaluating Praluent® were presented at the American Heart Association (AHA) Scientific Sessions. In November, the New England Journal of Medicine (NEJM) also published detailed results of this trial.
- Shan 6, a pediatric hexavalent vaccine, entered phase 3.
Phase 2:
- A phase 2 study evaluating the combination of isatuximab (anti-CD38 mAb) and cemiplimab (collaboration with Regeneron) in lymphoma was intitiated.
- A phase 2 study evaluating the combination of isatuximab and atezolizumab (PD-L1 inhibitor mAb) in solid tumors was initiated.
- A phase 2 study evaluating SAR440340 (an anti-IL33 mAb, collaboration with Regeneron) in atopic dermatitis was initiated.
- Positive primary analysis of the Phase 2b trial demonstrated the safety and efficacy of SP0232/MEDI8897 (anti RSV mAb - Respiratory Syncytial Virus, collaboration with Medimmune).
- Several projects in phase 2 were stopped:
- GZ389988, a TRKA antagonist, in osteo arthritis;
- ALX0171, an anti RSV nanobody (from Ablynx) for Respiratory Syncitial Virus;
- SAR425899, a GLP-1 / GCGR agonist, in obesity in type 2 diabetes patients;
- SAR407899, a rho kinase inhibitor, for microvascular angina;
Phase 1:
- SAR408701, an anti-CEACAM5, achieved positive proof of concept in a subgroup of lung cancer patients. A broad development program is expected to start by the end of 2019.
- BIVV001, a recombinant Factor VIII for Hemophilia A, achieved positive proof of concept with demonstration of sustained high factor levels at once-weekly dosing.
- SAR441000, a cytokine mRNA (collaboration withBioNTech AG) entered phase 1 in the treatment of melanoma.
- SAR443060/DNL747 (collaboration with Denali), an oral brain-penetrant small molecule (RIPK1 inhibitor), entered phase 1 clinical study in Amyotrophic Lateral Sclerosis (ALS) and Alzheimer’s disease.
- BIVV003, a Zinc Finger Nuclease (ZFN) gene editing technology issued from Bioverativ entered phase 1 in the treatment of sickle cell disease.
- SAR441344, an anti-CD40L mAb (license from ImmuNext), entered phase 1 in the treatment of multiple sclerosis.
- A next generation Pneumococcal Conjugate Vaccine (PCV) entered phase 1.
- Several projects in phase 1 were stopped:
- SAR439794, a TLR4 agonist immunomodulatory evaluated in peanut allergy;
- SAR247799, a S1P1 agonist evaluated in cardiovascular area;
- SAR438335, a GLP-1/GIP agonist in Type 2 diabetes;
- SAR228810, an anti protofibrillar AB mAb for Alzheimer disease;
- UshStat®, a myosin 7A gene therapy for Usher Syndrome 1B, will be discontinued contigent upon identification of out-licensing partner.
Collaborations
In January 2019, Sanofi and Regeneron announced a restructuring of their global Immuno-Oncology Discovery and Development Agreement for new IO cancer treatments. The 2015 agreement was scheduled to end in approximately mid-2020. This revision provides for ongoing collaborative development of two clinical-stage bispecific antibody programs (BCMAxCD3 and MUC16xCD3 bispecific). It also provides Sanofi with increased flexibility to advance its early-stage IO pipeline independently while Regeneron retains all rights to its other IO discovery and development programs.
In January 2019, BioNTech announced that it has extended its research collaboration with Sanofi initiated in late 2015 in the field of mRNA cancer immunotherapy.
In January 2019, MyoKardia, Inc. announced that it regained worldwide rights to all programs covered under its license and collaboration agreement with Sanofi. The collaboration has not been extended beyond the initial research term, which ended on December 31, 2018. As a result, MyoKardia now has regained global rights to all programs in its portfolio, including mavacamten (a Myosin inhibitor evaluated in obstructive and non-obstructive hypertrophic cardiomyopathy) and MYK-491 (a Myosin activator evaluated in dilated cardiomyopathy) and the license and collaboration will conclude in its entirety effective April 1, 2019.
In December Sanofi and Medicines for Malaria Ventures (MMV) agreed to transfer the operational responsibility for the development of Ferroquine/OZ439, to MMV in such a way that MMV would assume leadership while Sanofi remains the sponsor of the studies, fulfilling drug supply, regulatory and legal obligations. Ferroquine/OZ439 is a first in class combination for malaria previously developed in collaboration with MMV.
In November, Sanofi announced that it plans to collaborate with Denali Therapeutics Inc. on the development of multiple molecules with the potential to treat a range of neurological and systemic inflammatory diseases.
To access the full press release of the 2018 Q4 and full-year results, please click here.
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These statements include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects”, “anticipates”, “believes”, “intends”, “estimates”, “plans” and similar expressions. Although Sanofi’s management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include among other things, the uncertainties inherent in research and development, future clinical data and analysis, including post marketing, decisions by regulatory authorities, such as the FDA or the EMA, regarding whether and when to approve any drug, device or biological application that may be filed for any such product candidates as well as their decisions regarding labelling and other matters that could affect the availability or commercial potential of such product candidates, the absence of guarantee that the product candidates if approved will be commercially successful, the future approval and commercial success of therapeutic alternatives, Sanofi’s ability to benefit from external growth opportunities, to complete related transactions, and/or obtain regulatory clearances, risks associated with intellectual property and any related pending or future litigation and the ultimate outcome of such litigation, trends in exchange rates and prevailing interest rates, volatile economic conditions, the impact of cost containment initiatives and subsequent changes thereto, the average number of shares outstanding as well as those discussed or identified in the public filings with the SEC and the AMF made by Sanofi, including those listed under “Risk Factors” and “Cautionary Statement Regarding Forward-Looking Statements” in Sanofi’s annual report on Form 20-F for the year ended December 31, 2017. Other than as required by applicable law, Sanofi does not undertake any obligation to update or revise any forward-looking information or statements.