NEARLY HALF OF IBS PATIENTS SURVEYED REPORT THEIR IBS SYMPTOMS HAVE BEEN MORE CHALLENGING TO MANAGE IN THE PAST YEAR
Salix Pharmaceuticals Sponsored Second Annual Report, Patient Perspectives: Living with IBS Now
Report Findings May Help Health Care Providers Improve IBS Patient Outcomes
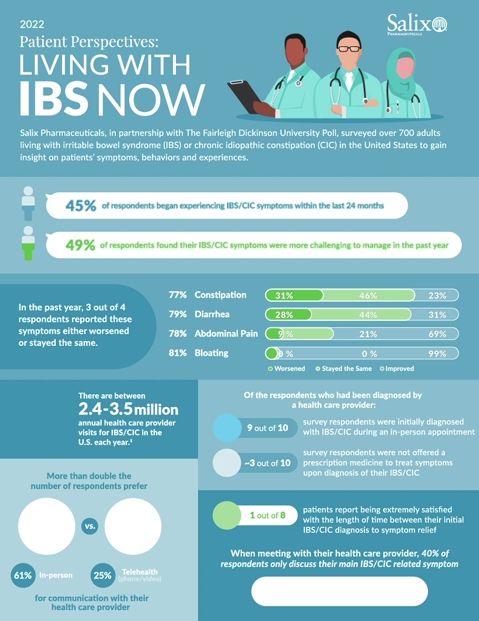
LAVAL, Quebec, April 4, 2022 – Bausch Health Companies Inc. (NYSE/TSX: BHC) (“Bausch Health”) and its gastroenterology (GI) business, Salix Pharmaceuticals (“Salix”), one of the largest specialty pharmaceutical companies in the world committed to the prevention and treatment of gastrointestinal diseases and disorders, today released the results from the second edition of its Patient Perspectives Survey, an annual survey of U.S adults living with irritable bowel syndrome (IBS) or chronic idiopathic constipation (CIC). The report, Patient Perspectives: Living with IBS Now, offers a current understanding of the behaviors and experiences of the IBS and CIC patient population in the past year (February 2021 – February 2022), including insights on diagnosis and symptom management. Most notably, almost half (49%) of respondents found their IBS or CIC symptoms have been more challenging to manage during the last 12 months. The report, which was developed based on a nationwide survey conducted in partnership with the Fairleigh Dickinson University Poll (FDU Poll), also addresses gaps in symptom management and reporting, and barriers to effective treatment. The survey also reveals trends in patient-provider communications.
“Salix remains committed to delivering impactful resources that inform the entire IBS and CIC community and address the needs of both patients and health care providers,” said Robert Spurr, president, Salix. “The findings of this year’s report illustrate a need for more meaningful health care provider visits to help improve clinical outcomes. As many as 3.5 million annual health care provider visits for IBS take place in the U.S. each year1, and it is our hope that this research will encourage productive dialogue and a collaborative approach to symptom management and guideline-based treatment options during these visits.”
Key findings from the report include:
.@SalixPharma’s second annual Patient Perspectives survey uncovers key insights about the experiences of adults living with IBS. Tweet
- Overall, 45% of patients acknowledged they began experiencing IBS or CIC symptoms within the last 24 months
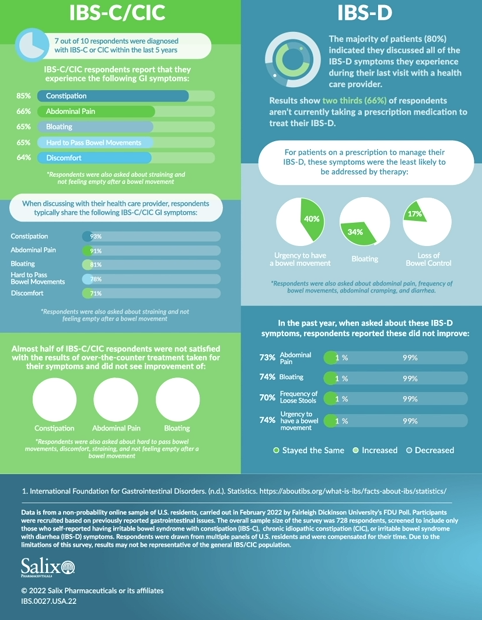
- More than three out of four patients surveyed report that several of their IBS or CIC symptoms included in the survey have not improved or have worsened over the last 12 months (between 77% and 81% of respondents)
- 92% of HCP-diagnosed patients surveyed were initially diagnosed with IBS or CIC during an in-person appointment with their health care provider
- More than half (61%) of patients chose in-person visits as their preferred method of communication with the health care provider who treats their IBS or CIC
- When meeting with their health care provider, 40% of patients shared they only discuss their main IBS or CIC-related symptom, rather than discussing all IBS or CIC-related symptoms
- Nearly one-third (29%) of patients were not offered a prescription medicine to treat symptoms upon diagnosis of their IBS or CIC
To view key insights from Salix’s Patient Perspectives: Living with IBS Now report, visit here. To view a comprehensive infographic, visit here.
About the Methodology Behind Patient Perspectives: Living with IBS Now
Data is from a non-probability online sample of U.S. residents and was carried out in February 2022 by Fairleigh Dickinson University’s FDU Poll. Participants were recruited based on previously reported gastrointestinal issues. The overall sample size of the survey was 728 respondents and was screened to include only those who self-reported having IBS with constipation, chronic idiopathic constipation or IBS with diarrhea. Respondents were drawn from multiple panels of U.S. residents and were compensated for their time (maximum $5.00). Due to the limitations of this survey, results may not be representative of the general IBS or CIC population.
Patient Perspectives: Living with IBS Now report
Patient Perspectives: Living with IBS Now report
About Fairleigh Dickinson University
Since 2001, Fairleigh Dickinson University’s FDU Poll has conducted survey research on issues of public importance. Utilizing best practices in survey methodology, FDU Poll produces authoritative statewide and national public polling. In addition, FDU Poll engages in research for corporate, non-profit and government entities. As a charter member of the American Association for Public Opinion Research’s Transparency Initiative, the FDU Poll is devoted to building public confidence in polling by embracing the open science of survey research.
About Salix
Salix Pharmaceuticals is one of the largest specialty pharmaceutical companies in the world committed to the prevention and treatment of gastrointestinal diseases. For more than 30 years, Salix has licensed, developed and marketed innovative products to improve patients’ lives and arm health care providers with life-changing solutions for many chronic and debilitating conditions. Salix currently markets its product line to U.S. health care providers through an expanded sales force that focuses on gastroenterology, hepatology, pain specialists and primary care. Salix is headquartered in Bridgewater, New Jersey. For more information about Salix, visit www.Salix.com and connect with us on Twitter and LinkedIn.
About Bausch Health
Bausch Health Companies Inc. (NYSE/TSX: BHC) is a global company whose mission is to improve people’s lives with our health care products. We develop, manufacture and market a range of pharmaceutical, medical device and over-the-counter products, primarily in the therapeutic areas of eye health, gastroenterology, and dermatology. We are delivering on our commitments as we build an innovative company dedicated to advancing global health. For more information, visit www.bauschhealth.com and connect with us on Twitter and LinkedIn.
Forward-looking Statements
This news release may contain forward-looking statements, which may generally be identified by the use of the words “anticipates,” “expects,” “intends,” “plans,” “should,” “could,” “would,” “may,” “believes,” “estimates,” “potential,” “target,” or “continue” and variations or similar expressions. These statements are based upon the current expectations and beliefs of management and are subject to certain risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. These risks and uncertainties include, but are not limited to, the risks and uncertainties discussed in the Bausch Health’s most recent annual report on Form 10-K and detailed from time to time in Bausch Health’s other filings with the U.S. Securities and Exchange Commission and the Canadian Securities Administrators, which factors are incorporated herein by reference. They also include, but are not limited to, risks and uncertainties caused by or relating to the evolving COVID-19 pandemic, and the fear of that pandemic and its potential effects, the severity, duration, and future impact of which are highly uncertain and cannot be predicted, and which may have a material adverse impact on Bausch Health, including but not limited to its project development timelines, and costs (which may increase). Readers are cautioned not to place undue reliance on any of these forward-looking statements. These forward-looking statements speak only as of the date hereof. Bausch Health undertakes no obligation to update any of these forward-looking statements to reflect events or circumstances after the date of this news release or to reflect actual outcomes, unless required by law.
References
- International Foundation for Gastrointestinal Disorders. (n.d.). Statistics. https://aboutibs.org/what-is-ibs/facts-about-ibs/statistics/
SAL.0024.USA.22
©2022 Salix Pharmaceuticals or its affiliates.
Investor Contact:
Arthur Shannon
[email protected]
(514) 856-3855
(877) 281-6642 (toll free)
Media Contact:
Lainie Keller[email protected]
(908) 927-1198
IBS.0024.USA.22 V2.0